PubChem is a reliable source of chemical information from authoritative sources. You can look up element names, symbols, atomic masses and more, or test your knowledge with a periodic table game. You will see in the next chapters that the chemical properties of elements are determined by the number of valence electrons. These rare earth metals are 2 periods behind because the f electrons are even higher in energy than the d electrons. PubChem Periodic Table of Elements is an interactive tool that lets you explore the properties and trends of chemical elements. We can see from the electron configuration of a carbon atom-1 s 22 s 22 p 2-that it has 4 valence electrons (2 s 22 p 2) and 2 core electrons (1 s 2). You can use the periodic table to help you determine how many valence electrons an element (specifically, a neutral atom of. It is located on the outermost shell (in this case, the shell resembles a ring). Valence shell electrons (or, more simply, the valence electrons) are the electrons in the highest-numbered shell, or valence shell, while core electrons are the electrons in lower-numbered shells. For example, in the figure below showing a simplified diagram of sodium’s electron configuration, the valence electron is shown in red. Thus, it is convenient to separate electrons into two groups.
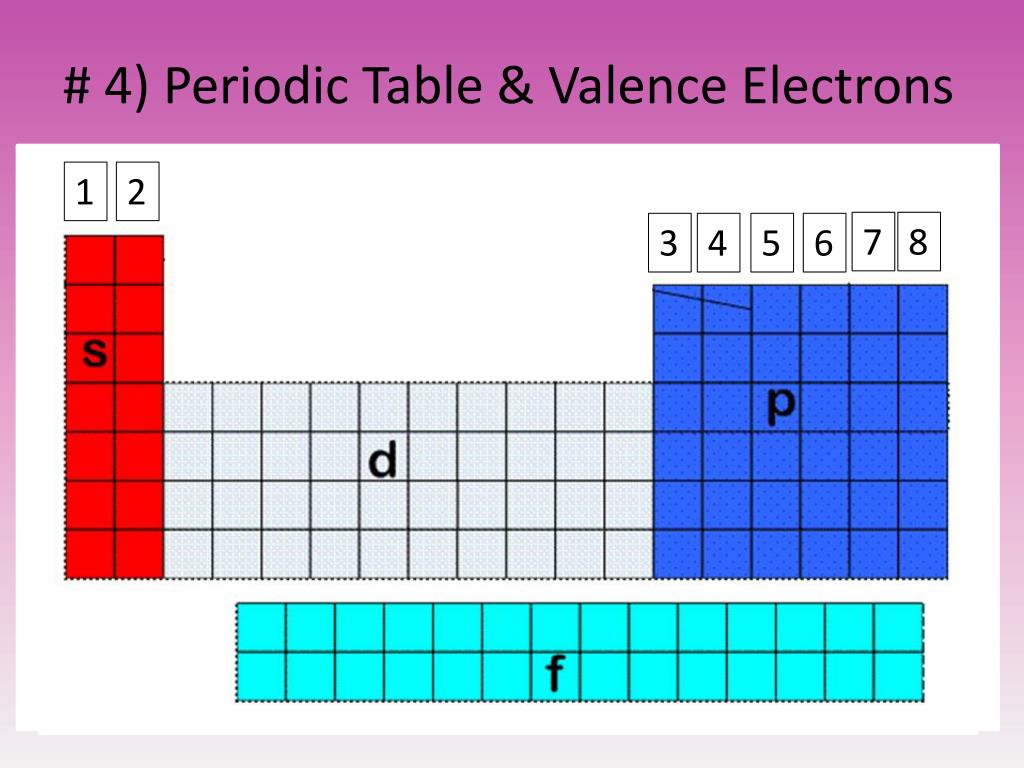
Now, for the sake of this video, Im gonna focus most on the extremes of the periodic table, the groups at the left and the right. The path a specific element will take depends on where the electrons are in the atom and how many there are. And because elements with similar valence electrons will have similar reactivities, they will form similar ions. \,3s^1 \nonumber \]Ī chemical reaction results from electron removal, electron addition, or electron sharing of the valence electrons of the different atoms.
